
HOME > Research
Human Clinical Study Ⅰ(9):
Kobayashi et al. (1991) asked 16 terminal cancer patients (Table 2) to consume S100 Milk tablets (3-18 tablets, about 0.3g per tablet) for two months, and then they evaluated the satisfaction index of the patients during the study and the changes in the clinical index shown by the QQL (Quality of Life) scale. After taking the S100 tablets for one month, 30.8% of the patients felt that the milk tablets were helpful and 15.4% of the patients thought their bodies were significantly improved. After taking for two months, up to 50.0% of the patients thought their bodies were significantly improved. The overall improvement rate was as high as 60% (Fig. 7)(9).
Improvements in clinical symptoms due to cancer included: 37.6% of the patients thought the symptoms of abdominal distension and pain were relieved; 31.3% thought that systemic lassitude was improved; 26.7% thought that constipation was reduced; 25.1% thought the feeling of nausea and vomiting was reduced; 25% thought conditions including high fever, sweating and loss of appetite were improved(9).
| What kind of cancer do subjects suffer from? | ||||
| Name of diagnosis | Subjects | |||
| Breast cancer | 8 | |||
| Gastric cancer | 1 | |||
| Uterine cancer | 1 | |||
| Esophageal cancer | 1 | |||
| Renal cell carcinoma | 1 | |||
| Rectal cancer | 1 | |||
| Lung cancer | 1 | |||
| Pancreatic cancer | 1 | |||
| Chondrosarcoma | 1 | |||
| Total | 16 | |||
| Table 2. Cancer types of 16 terminal cancer patients(9). | ||||
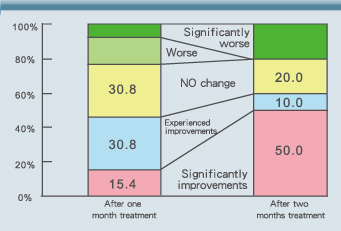
Fig. 7 After taking S100 Milk tablets for one month, 30.8% of the subjects thought they experienced improvements; 15.4% thought the improvements were significant. After taking for two months, 10% of the subjects felt there were improvements; 50% thought there were significant improvements(9).
There were also improvements shown in blood chemistry indexes. The levels of RBCs, Hb and Hct in subjects all increased significantly after they took the S100 Milk tablets reinforcing what was reported by the subjects themselves(9).
Human Clinical Study Ⅱ(10) :
We understand that regular exercise is beneficial for overall health. However, prolonged and intense physical activity may have adverse effects on the body, potentially leading to a decrease in immunity, induction of inflammatory responses, and subsequent harm to muscles and organs. It may also result in gastrointestinal symptoms such as bloating, abdominal pain, diarrhea, vomiting, and erosive gastritis.
In a clinical human trial conducted in Japan (Ma et al., 2020), researchers recruited seven healthy young individuals who were randomly divided into two groups. One group consumed immune protein powder (IMP), while the other group consumed regular protein powder (placebo protein, PLA). Participants ingested the assigned product twice daily, with each serving containing 10 grams, for 8 weeks. Following this period, a 1-week washout period was implemented, and then the groups switched products for an additional 8 weeks. Before product use (M0), after 1 month of product use (M1), and after 2 months of product use (M2), participants underwent a 3000-meter sprint test, and relevant indicators were measured.
Intestinal Fatty Acid Binding Protein (I-FABP) is an indicator used in hospitals to assess intestinal damage. The experimental results demonstrated a significant increase in I-FABP after intense exercise, indicating damage to the intestines. However, the group supplemented with IMP showed a significant reduction in the increase of I-FABP caused by intense exercise (refer to Fig. 8). This confirms that immune protein powder effectively protects intestinal health during strenuous physical activity(10).
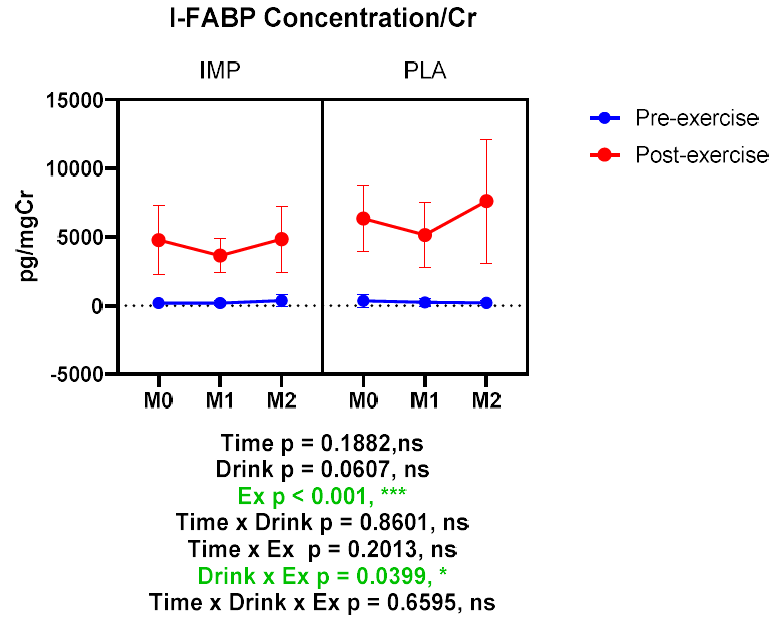
Fig. 8. I-FABP concentration corrected for creatinine. Data are expressed as mean ± standard deviation.
* p < 0.05 和*** p < 0.001. Green words indicate significance<0.05 (10).

